“Our response was too orientated toward the management of previous outbreaks,” explained Jean-Hervé Bradol, the director of Médecins Sans Frontières’ (MSF; Doctors Without Borders) internal review body, the Centre de Réflexion sur l’Action et les Savoirs Humanitaires (CRASH). “We wasted time before speaking about a vaccine and treatments. It’s very hard to imagine controlling this epidemic now without a vaccine” (Flynn and Bartunek 2014).
In an outbreak, public health is haunted by the specter of belatedness. Delays in diagnosis reduce survival rates; sluggish case-detection redoubles contagion. Time’s ravages are materialized by a single equation: the reproductive ratio, or R0, which determines the average number of people a sick individual will infect. Spreading only through close physical contact with very sick people or corpses, Ebola’s R0 is not of an apocalyptic order. Its virulence, while terrifying, is ultimately self-limiting, as patients routinely die before they can infect many others. Reducing transmission is thus relatively straightforward: the sick must be isolated, their contacts monitored, and the dead safely buried. In the 24 known outbreaks recorded since the virus was first identified in 1976, these approaches have kept the numbers of infected lower than 200 on average.
Needless to say, the current situation in West Africa is different. A year now from the first or “index” case, the disease has “ping-ponged” from village to city and back, moving into new districts, spilling across borders, and boarding planes. The reasons for this outbreak’s magnitude—approaching 18,000 cases and well over 6,000 reported deaths (as of early December 2014)—are both structural and contingent: the density and mobility of the population; an endemic distrust of the government; perilously weak health systems; the underfunding of the World Health Organization (WHO) and an excessive reliance on MSF; the death of a high-profile imam; the siting of a treatment center; the decision to not give a well-known local doctor access to experimental treatment, etc. Whatever the reason, the outbreak’s pace is now a feature of its own scale. Dragging the R0 down will demand population-level methods.
Protective span is vaccine territory, and there are currently several candidates in the pipeline, two of which are in advanced stages of development. GlaxoSmithKline’s (GSK) cAd3-ZEBO vaccine, involving a non-replicating chimpanzee adenovirus, has been shown to be safe in healthy volunteers and is on track for large-scale clinical trials among afflicted populations as early as January 2015. An alternative vaccine, rVSV-ZEBO, developed by the Canadian company NewLink Genetics in collaboration with Merck and based on a weakened version of the vesicular stomatitis virus (VSV), is not far behind. While the details of trial design, locations, and target populations are still being worked out, these investigations will be supported by MSF, which will provide trial locations, funding, and clinical oversight.[1]
The paradigmatic example of biomedical triumphalism, a successful vaccine offers the promise of a way of tackling the current outbreak and a permanent solution for those yet to come (compare Rees 2014). But while expectations are high, immunization is not the only method on trial. In a seeming radical reversal of protocol, MSF and other international and nongovernmental aid organizations are emphasizing community-based responses, involving the distribution of home disinfection kits and the construction of Ebola care units staffed by non-expert volunteers.
What MSF describes as “unprecedented and imperfect measures” clearly belong to a different “political economy of hope” than the Ebola-free futures projected by the cAd3-ZEBO and VSV-ZEBO vaccines (MSF 2014; Novas 2006). And yet, vaccines and community-based methods share an impetus to re-create the temporality of control, to restore a “response time” that seems increasingly out of joint. “Going forward we are now hunting the virus, chasing after the virus,” said WHO Director-General Margaret Chan said in a recent interview (Mazumdar 2014). As long as the outbreak is able to “run ahead” of efforts to contain it, she noted, the “risk to the world is always there” (Mazumdar 2014). Through a series of compromises and hedges between care and experimentation, a complex technical assemblage of humanitarian biomedicine is now in operation, an assemblage that attempts to simultaneously address individual survival and global security (Lakoff 2010; Redfield 2012).
Pre-Presentation
A quick “burnout” was what MSF—de facto the only organization capable of managing viral hemorrhagic outbreaks—had expected. But when Ebola cropped up in cities—Conakry (Guinea), Monrovia (Liberia), and finally Freetown (Sierra Leone)—tracking and isolating individual cases became impossible. Well-equipped and expert-staffed treatment centers were overwhelmed and, in some cases, functioned only as spaces of quarantine. Those who sought treatment traveled further in search of quality care, potentially infecting others along the way. Many others came to associate clinics with death and chose to stay at home, avoiding contact with health services altogether.
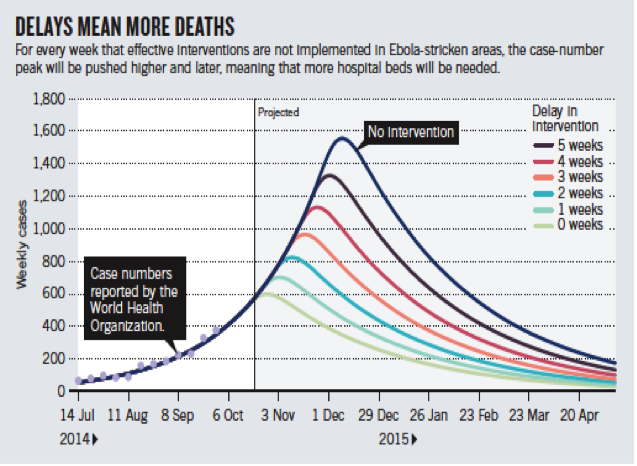
When the international response finally kicked into gear it established an ambitious benchmark: isolating 70% of cases by December 1, 2014, and 100% by the beginning of 2015.[2] To that end, foreign governments pledged human and material resources to build and staff new centers. From the start, however, these efforts were caught in a game of catch-up, chasing exponential projections that placed the number of beds perpetually behind the curve (Figure 1).
Again, the issue here is timing. It is not enough to isolate patients: they must be secluded before they are infective (within three days of the first symptoms, the models suggest). Slowing transmission thus demands expanding the biomedical frontier from centers of expertise to the uncertain realm of the “community.”
Community Care Units (CCUs) provide that extension. Drawing on a concept of “community care” originally developed within the UK National Health Service in the 1960s (Draper 1967), Ebola CCUs are now being erected in the outskirts of villages and periurban neighborhoods all across the outbreak area, using tents, tarps, makeshift materials, and repurposed buildings. CCUs can provide anywhere from 10 to 30 beds and are supported by unspecialized and minimally trained volunteers from the community—ideally survivors—using equipment and supplies donated by humanitarian agencies. Clean water, sanitation, and food and basic medicine are provided; care is primarily palliative. Diagnosis or any other interventions that requires drawing blood is not among the services offered. Because Ebola’s early-stage symptoms are considered relatively less serious, a single health worker is expected to manage several patients at a time.
 The number of CCUs has grown rapidly; the UK government plans to build at least 200 more. Guidelines are still being hammered out. As Chris Whitty and colleagues put it, “optimal design will be learned on the fly. We know how to minimize infection in dedicated Ebola wards with highly trained and supervised staff, but not in facilities with lightly trained staff in which most people do not have Ebola (but some do)” (2014:194).
The number of CCUs has grown rapidly; the UK government plans to build at least 200 more. Guidelines are still being hammered out. As Chris Whitty and colleagues put it, “optimal design will be learned on the fly. We know how to minimize infection in dedicated Ebola wards with highly trained and supervised staff, but not in facilities with lightly trained staff in which most people do not have Ebola (but some do)” (2014:194).
While uncertainties remain regarding the quality of care and the risks involved in its delivery, CCUs are not conceived as ends in themselves, but rather as instruments of triage: a waiting post until patients whose cases are confirmed can be sent to a proper treatment facility. Within this framework, even the distribution of personal protective equipment to households is not seen as a last-ditch humanitarian effort, but rather as another link in a chain that will eventually lead one to the expert care provided by MSF (Leach et al. 2014).
The key selling point of the CCU is its proximity to the community, in space and in spirit. If this outbreak has taught us anything, it is that disease control is not merely a question of access but also of acceptability; until quite recently, the response has been perilously short on both. By enfolding the grassroots into health care delivery, the CCU provides a stopgap to containment efforts, but only if patients regard these places as sites where they will receive quality care. Things will fall part, as one MSF logistician put it, “if people feel that they are being left with scraps, while Europeans are treated with ZMAPP.”
In short, CCUs operate under the expectation that biomedical resources—in the form of cures, diagnostics, staff, and support—are at hand. Of course, with no drug or vaccine yet available and with a patient fatality rate around 60%, the promise of care feels shallow. “Given the risks that health workers take, getting Ebola vaccines to staff working in the units as soon as they have proved safe is an ethical imperative” (Whitty et al. 2014:194).
Speeding Vaccines
Incredibly, that time might not be far off. International consortia involving academic, government, and commercial partners have been quickly assembled to design and implement fast-track clinical trials; regulatory requirements have been streamlined; plans have been put forward to set up an indemnity fund to insure pharmaceutical companies if the vaccines prove to have any dangerous side effects, and the United States has provided immunity against any legal claims (Federal Register 2014). Having shrunk the timeline from the usual years to months, preparations have begun for large-scale studies in Ebola-affected countries to begin in January 2015.
While logistical questions remain—ranging from the number of doses needed to the cold-chain requirements for vaccine delivery—the most pressing concern is which population to target. Prioritizing “frontline workers”—those clinicians, contact tracers, burial teams, and volunteers providing community-based care—is the obvious strategy: in addition to being at greater risk of infection, these groups are arguably more capable of understanding the risks of an experimental vaccine. Informed consent, it is reasoned, will be more genuine and the potential backlash vaccines often create—for example, rumors about sterilization—will be minimized (e.g. Feldman-Savelsberg et al. 2000; Kaler 2009). Whether or not foreign health volunteers should be included in that cohort is unclear.
Another possibility being considered is a “ring vaccination” approach, like the one used during the WHO campaign to eradicate smallpox. This involves containing new foci of infection, targeting primary or secondary contacts of the infected, and following the importation of the disease to new countries. While the data suggest that while the fatality rate is much higher for those over the age of 45, it is likely that people will want their children were prioritized for vaccination.
In any case, selecting inclusion criteria is not merely a matter of determining who would benefit the most if the vaccine should prove successful. The explicit motivation for accelerating development is that a vaccine can address this outbreak. MSF’s Jean-Hervé Bradol’s admonitions express a frustration: if clinical trials had started earlier, transmission could have been halted and lives saved.
But regardless of the impact a vaccine would have on transmission ongoing in West Africa, the outbreak offers a scientific opportunity. “This must be the last time we are taken by surprise” was a refrain at the WHO Ebola Vaccine Consultation back in September. A vaccine has the unique capacity to act as bulwark against the unforeseen. Indeed, while the industry has capacity to produce vaccines in bulk, by the time trials are concluded, the dent a large-scale immunization program would make in the current West Africa epidemic would be relatively small. Rather, development is driven by the “inevitability” of future outbreaks: “All efforts to develop, test, and approve vaccines must be followed through to completion at the current accelerated pace…as a contribution to global health security, fully licensed and approved vaccines should be stockpiled in readiness for the next Ebola outbreak” (WHO 2014).
For pharmaceutical companies, government stockpiles would certainly mitigate the financial risks of developing a drug for which the poor cannot pay. However, to achieve this goal, companies must be sure that they have clear indications of vaccine efficacy. It is still unclear, for instance, how acceleration will affect the quality of the data required for licensure; the position of the U.S. Federal Drug Administration (FDA) on the evaluation of these “fast-track” trials is still open.
It is precisely at this point where the humanitarian and biosecurity potentials of the vaccine come into conflict. The FDA has so far been emphatic that randomized, double-blinded, placebo-controlled clinical trials (RCTs) are necessary for licensure. MSF, however, has put up strong opposition to a trial design that would mean denying potentially effective protection to those most at risk. While arguably there is genuine uncertainty about the benefits of these vaccines, the promise of the early-stage studies and the scale of the current outbreak have pitted concerns about investigative rigor against ethical imperatives of equity and compassion. The political consequences of these experiments are further amplified by fraught histories of mass immunization and iatrogenesis in Africa (e.g., Lachenal et al. 2010; Feldman-Savelsberg et al. 2000; Moulin 1996). As a representative from a West African regulatory authority put it: “For the African community we must have high quality protection that satisfies international standards, not rushed African standards (WHO, Consultation September 28th, 2014).”[3]
To enact this balance between investigative integrity and humanitarian compassion, between accelerated access and unnecessary exposure, a few strategies are being pursued. First, RCTs of safety and immunogenicity will take place among Africa populations in “at-risk settings,” but not where there is currently high transmission: for instance, Mali, Cameroon, or Ghana. These investigations will generate data on dosage and safety, adverse reactions, and reactivity with HIV/AIDS or other compromising infections. Almost simultaneously, head-to-head vaccine trials, comparing the two candidates potentially with a booster, will be rolled out in affected populations using an adapted randomized schedule referred to as a “stepped wedge” design (Figure 2). This will involve vaccinating groups in a sequence over an extended period of time (probably about a month): everyone will get vaccinated, but some before others. A stepped wedge has previously been used in situations where the intervention on trial is believed to do more good than harm or, alternatively, where logistical reasons (e.g., a limited supply) makes a simultaneous roll-out of the intervention to all participants difficult (compare Halloran 2010). Such a design presents obvious challenges, however, as the lag time might introduce new bias. What if virus mutates, for instance, or the incidence of other diseases (such as malaria) rises and interferes with the vaccine’s immunological response? Yet staggering the intervention into steps provides an opportunity to stop the trial if the vaccine proves to be ineffective. It creates the space to experiment within an emergency, to balance countervailing norms of access and evidence.
Protocols are still being finalized; the questions these experiments raise are legion. At the WHO back in September, an official in the US Department of Health and Human Services (HHS) emphasized that “time is not on our side.” Quick decisions had to be made about the dosage level and the inclusion of vulnerable populations (e.g., patients with HIV, pregnant women) in a situation where very little is known about what immune response will be good enough to ensure protection. Experimental vaccines and therapies should be made available but without any clear sense of their efficacy. Would populations storm experimental sites or run from them in fear? “This is a Kafkaesque situation,” the doctor conceded. It seemed a perfect epithet for the compulsion and deferral that characterizes humanitarian experimentation.